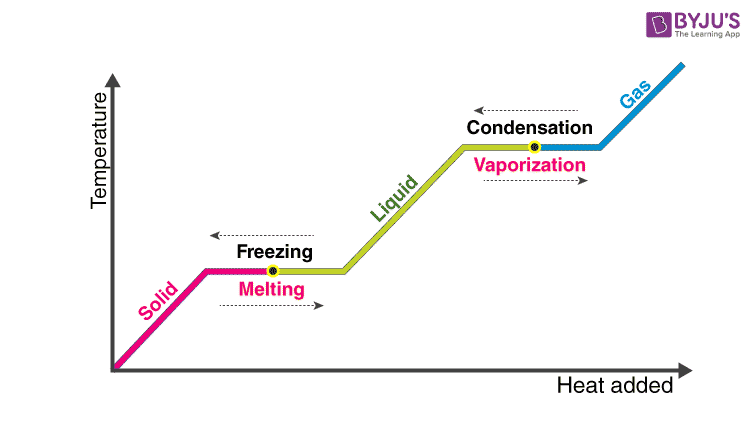
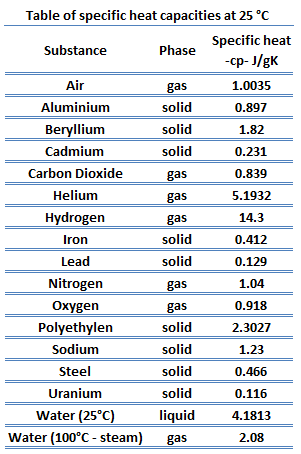
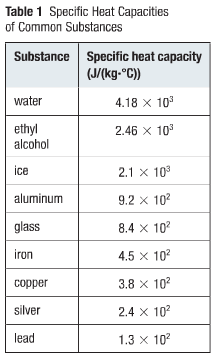
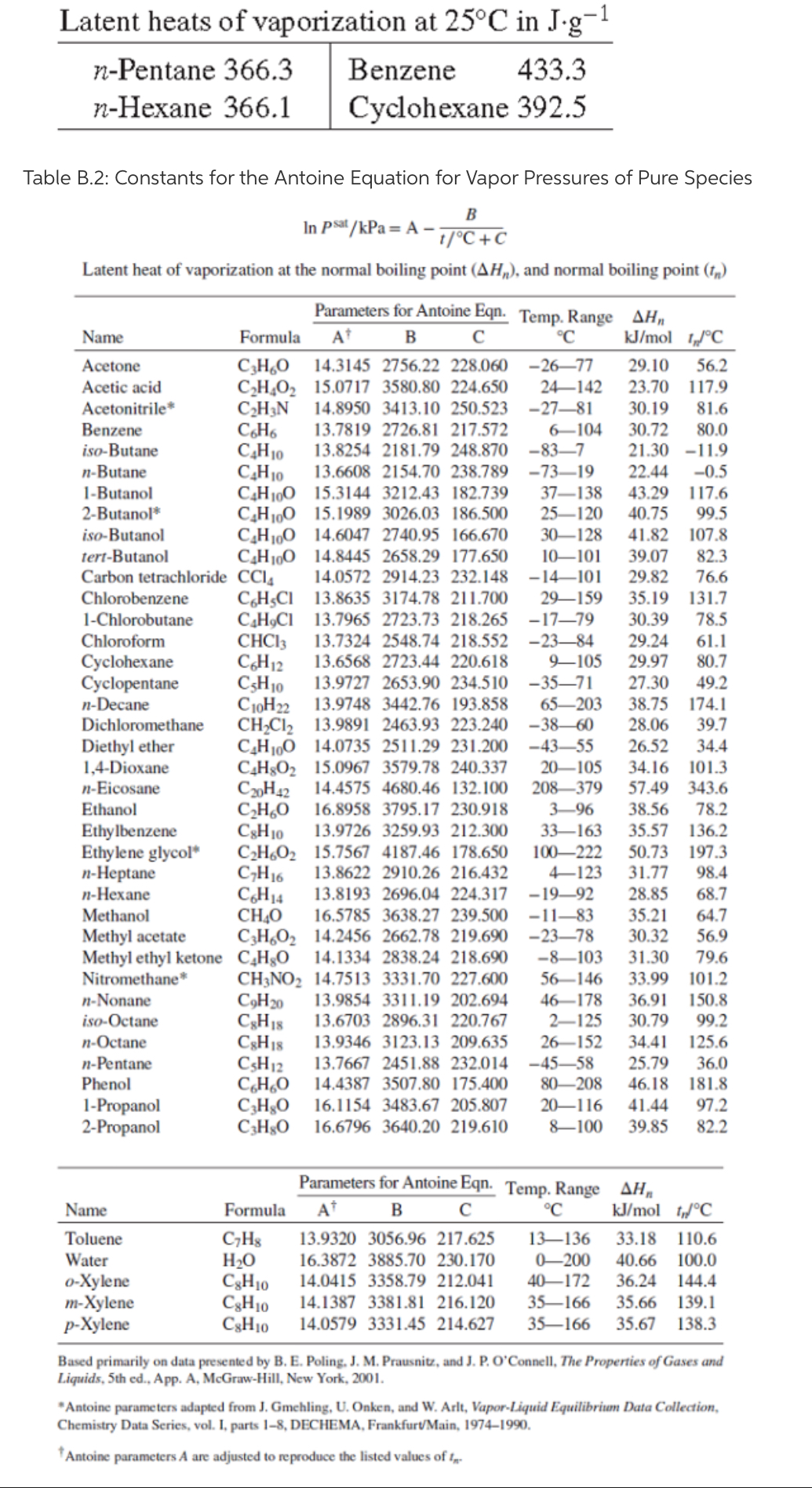
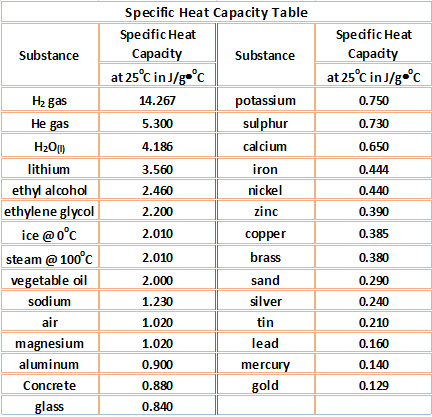
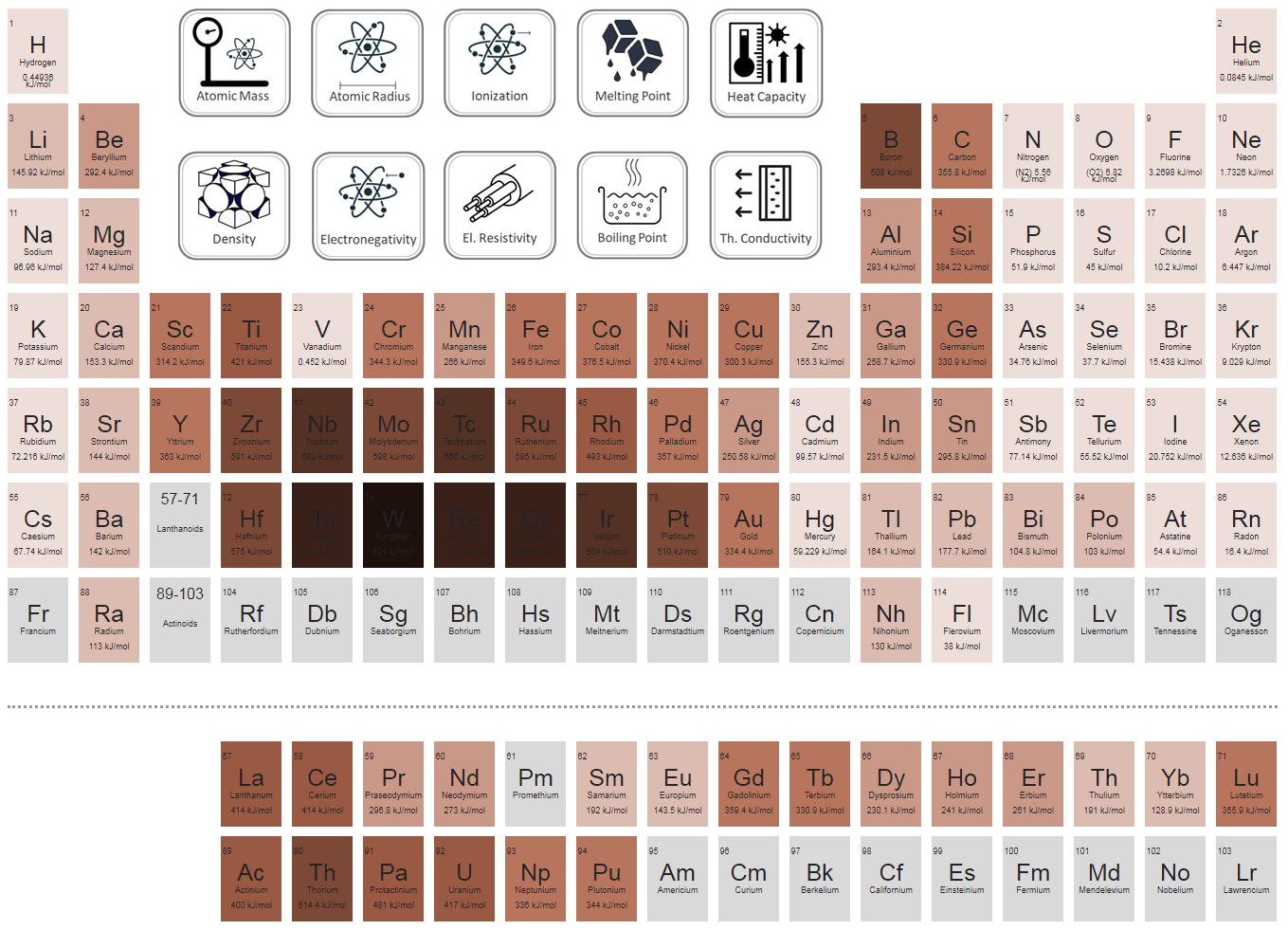
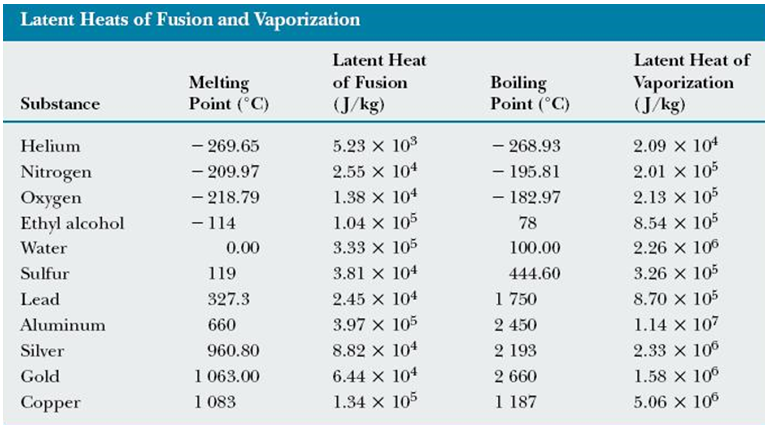
Reducing the experimental error in an experiment to determine the latent heat of vaporization of liquid nitrogen
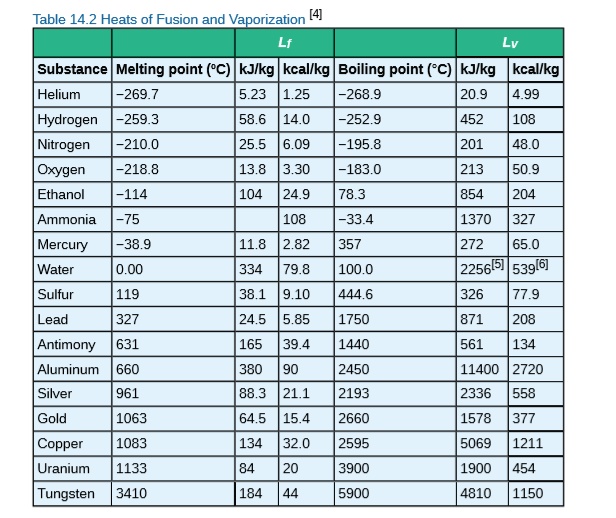
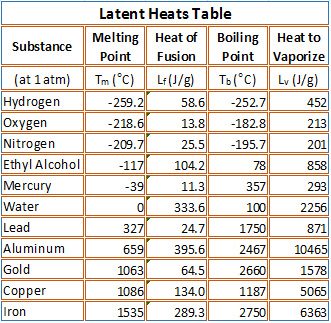
SOLVED: Table 14.2 Heats of Fusion and Vaporization [4 Substance Melting point ('C) kJlkg | kcallkg Boiling point (FC) kJlkg kcallkg Helium 269. 5.23 1.25 268.9 20.9 Hydrogen -259.3 58.6 14.0 -252.9


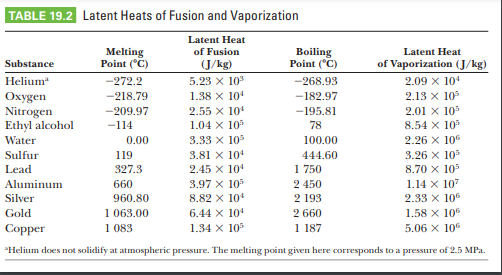






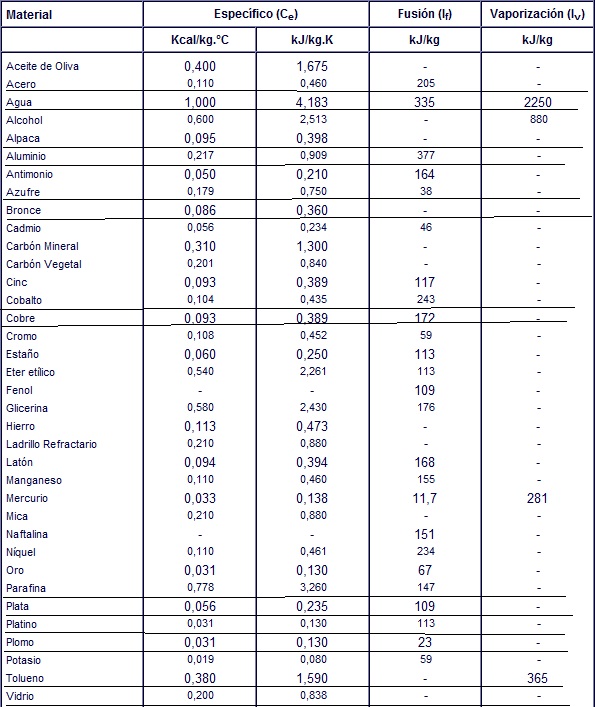
![PDF] Latent heat of vaporization for selected foods and crops | Semantic Scholar PDF] Latent heat of vaporization for selected foods and crops | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/e5e36a21914ce3ba4f27cd724afeb4e94d98ddce/4-TableII-1.png)