Question Video: Calculating the Concentration of H⁺ Ions Given the Acid Dissociation Constant and Degree of Dissociation | Nagwa

In the cubic crystal of CsCl (d = 3.97 g cm^(-3)), the eight corners are occupied by Cl^(Θ) with a Cs^(o+) at the centre and vice versa. Calculate the distance between the
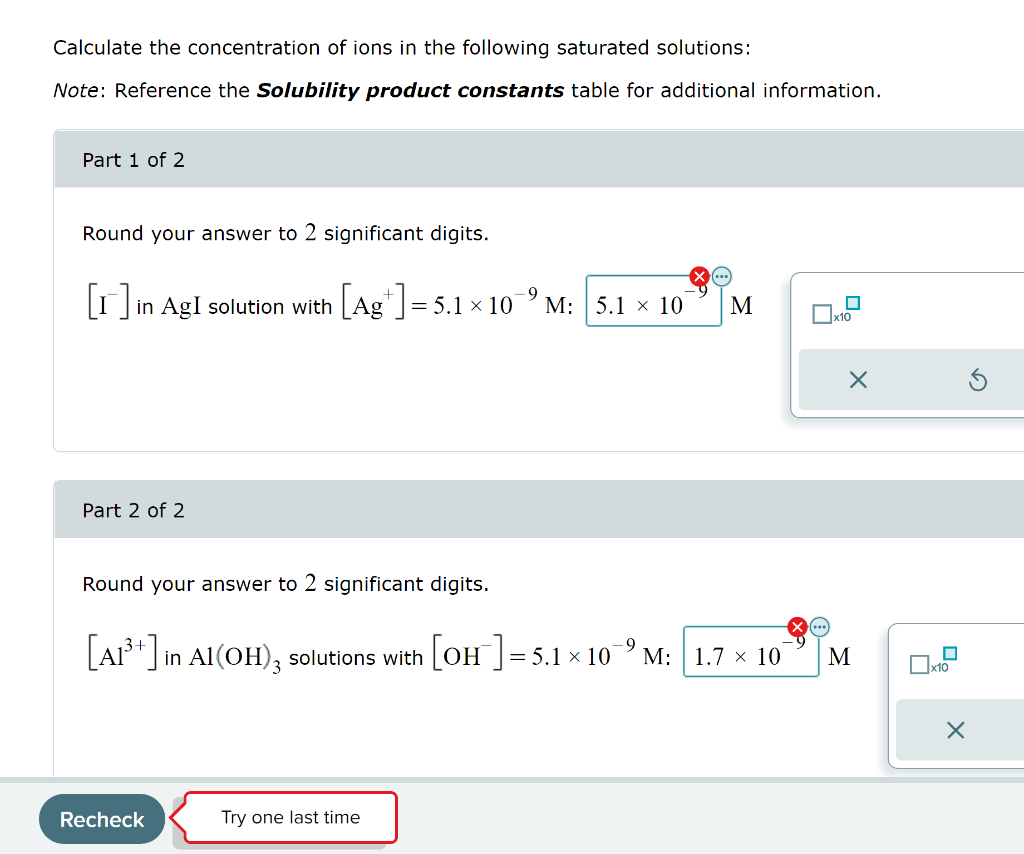
![SOLVED:Calculate the concentration of ions in the following saturated solutions: (a) [I^-] in AgI solution with [Ag^+]=9.1 ×10^-9 M (b) [Al^3+] in Al(OH)3 solution with [OH^-]=2.9 ×10^-9 M SOLVED:Calculate the concentration of ions in the following saturated solutions: (a) [I^-] in AgI solution with [Ag^+]=9.1 ×10^-9 M (b) [Al^3+] in Al(OH)3 solution with [OH^-]=2.9 ×10^-9 M](https://cdn.numerade.com/previews/80f388f1-f5ed-49d4-8950-f7804b8fe1f3_large.jpg)