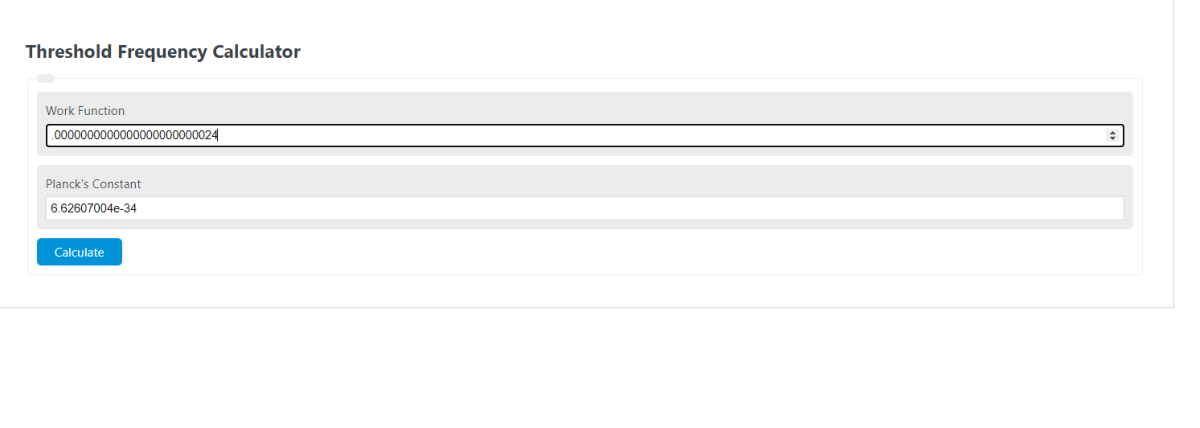
Electrons are emitted with zero velocity from a metal surface when it is exposed to radiation of wavelength 6800 ∘A . Calculate threshold frequency v0 and work function w0 of the metal.
calculate the threshold frequency of the metal from which the photoelectrons are emitted with zero velocity when exposed to radiation of wavelength 6800 Armstrong

The threshold frequency v0 for a metal is 7.0 × 10^14 s ^-1. Calculate the kinetic energy of an electron emitted when radiation of frequency v = 1.0 × 10^15 s^-1 hits the metal.

CHEM 101: Photoelectric Effect - Threshold Frequency and Binding Energy from Kinetic Energy - YouTube

a) Calculate the energy of an excited state for hydrogen.(b) Define threshold frequency of work function.(c) Work function for C5 atom is 1.9eV. Calculate kinetic energy for photoemitted electron if C5 metal

Calculate the threshold frequency of photon for photoelectric emission from a metal of work - YouTube

Calculate the threshold frequency of a photon from a photosensitive surface of work function 0.2 eV . Take h=6.6xx10^(-34)Js.

Calculate the threshold frequency of the metal from which the photoelectrons are emitted with zero velocity when exposed to radiation - Chemistry - - 14932835 | Meritnation.com