information theory - How to calculate conditional entropy using using this tabular probability distribution? - Mathematics Stack Exchange

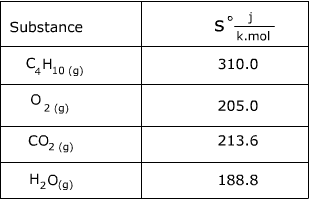
Calculate the entropy of Br2(g) in the reaction H2(g) + Br2(g) ⟶ 2HBr(g), S^o = 20.1 JK ^-1 given entropy of H2 and Hbr is 130.6 and 198.5 J mol^-1K^-1













![15.2/R1.4 Calculate the standard entropy change for a reaction [HL IB Chemistry] - YouTube 15.2/R1.4 Calculate the standard entropy change for a reaction [HL IB Chemistry] - YouTube](https://i.ytimg.com/vi/IwRy4iYVQLI/maxresdefault.jpg)