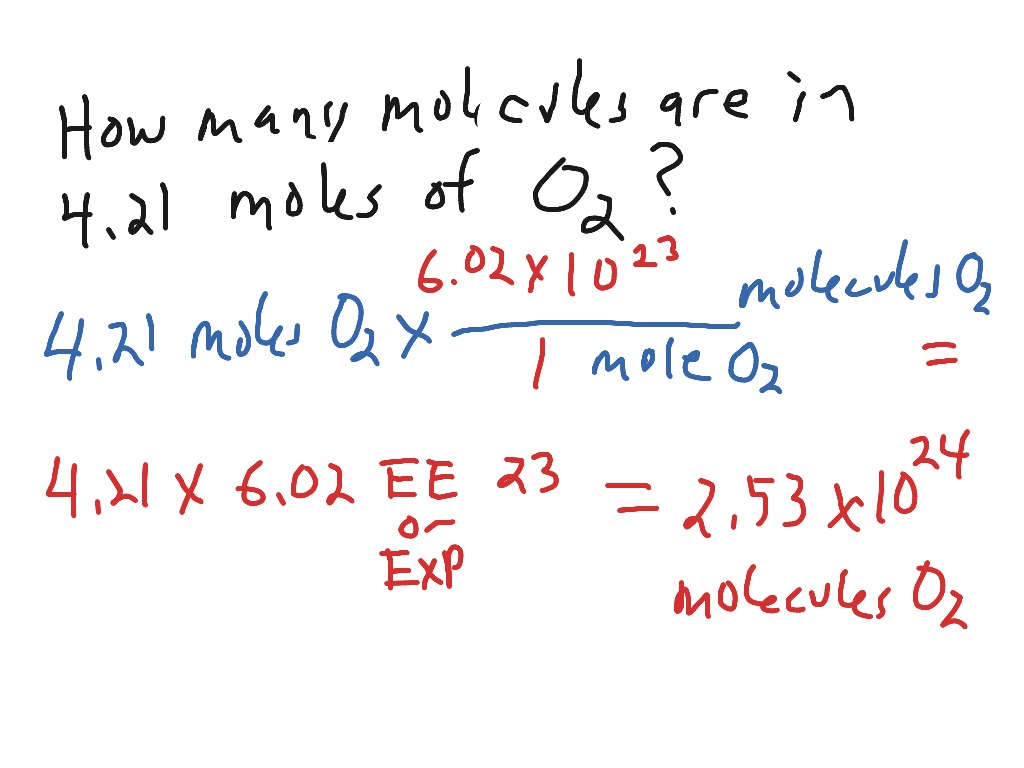How to calculate the number of molecules of an element in a compound if I only know the total mass - Quora

Calculate the number of molecules of sulphur (S8) present in 16 g of solid sulphur (Aton mass of S = 32 )
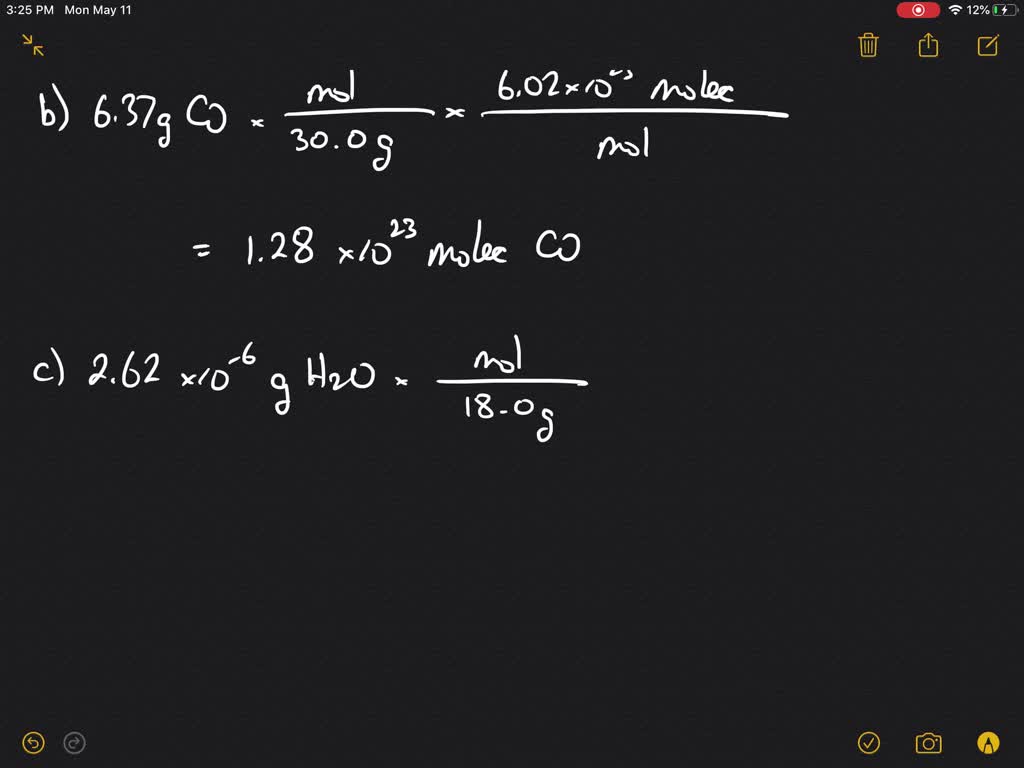
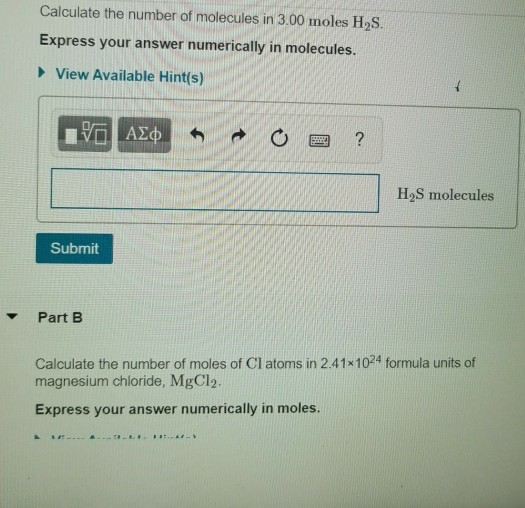
SOLVED:Calculate the number of molecules present in each of the following samples. a. 6.37 mol of carbon monoxide b. 6.37 g of carbon monoxide c. 2.62 ×10^-6 of water d. 2.62 ×10^-6
Calculate the number of particles in each of the following: (a) 48 g of Mg (b) 8 g of O2 (c) 0.1 mole of carbon (Atomic mass Mg = 24 u, O =

Calculate the number of particles in each of the following: (i) 46 g of Na atoms (number from ma... - YouTube

Calculate number of molecules in 3.6 g of water. | 10 | MOLE CONCEPT AND STOICHIOMETRY | CHEMIST... - YouTube